Lifesciences & Healthcare
Helped save over 30 million vaccines
Saving lives of 20,000+ babies in India every year
Plusstainibility at Work

Plusstainability to us is to channelize the concepts of thermal energy in promoting good health and wellbeing for the most vulnerable sections of the society. Thermal energy when stored in Phase Change Materials provides energy access for essential newborn care as well as for temperature sensitive lifesaving drugs and vaccines.
Pluss pre-validates its solutions using Smart CAE(computer aided engineering) – a cutting-edge software adapted to simulate performance of thermal packaging solutions equipped with phase-change materials (PCMs) enabling over 80% reduction in energy, fuel and wastage of packaging material on developmental cycle.
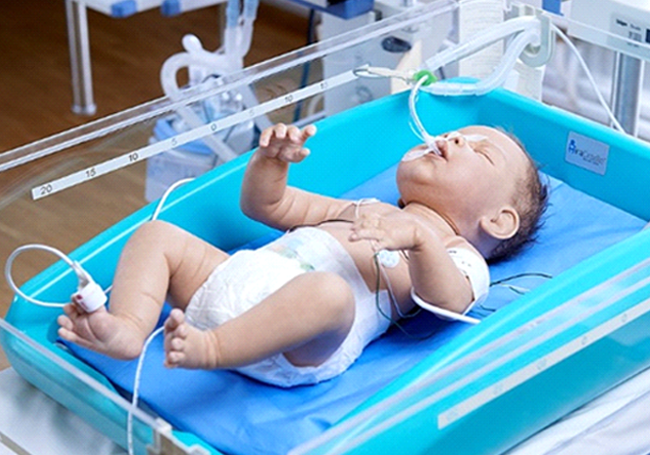
Essential NewBorn Care

More than 650,000 newborns die every year of birth asphyxia, second largest cause of neonatal deaths, and 90% of these deaths are in developing countries with deliveries made in low resource setting. Therapeutic hypothermia, which is controlled cooling at 33.5° C, is the only clinically proven method to treat asphyxiated babies. PLUSS’ patented and award-winning device, MiraCradle® Neonate Cooler, made cooling simpler, efficient and ten times more affordable than the standard equipments. Based on advanced Phase Change Material (PCM) technology the device is safe, validated, CE approved, non-electric and reusable, giving healthcare access to those who need it the most.
MiraCradle® at Work
• Saving lives of 20,000+ babies in India every year and deterring the onset of debilitating conditions like autism and cerebral palsy.
• Only clinically proven system for treating birth asphyxia by inducing therapeutic hypothermia even in low resource countries
• World’s most affordable system based on patented Phase Change Material (PCM) technology.
• Installed at over hundreds of hospitals in India and worldwide
Optimizing Vaccine Efficiency
Studies estimate that over 30% vaccines are
wasted every year for cold-chain issues.
Freezing of vaccines and potency loss
Celsure® Temperature Controlled
Packaging for Pharmaceuticals
and
Biological Products

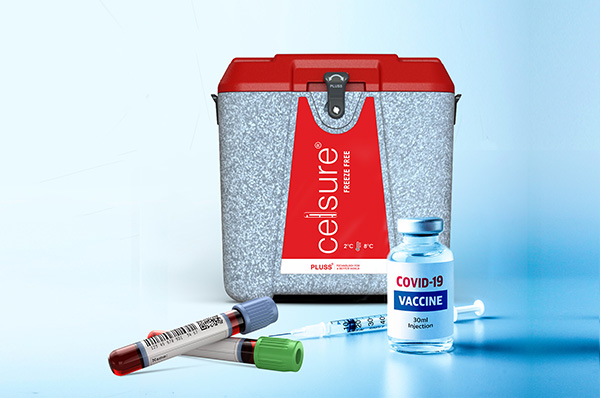
Celsure® Temperature Controlled
Packaging Benefits

- Helped save over 30 million vaccines and counting
- Provides precise temperature between 2° to 8° C, 15° to 25°C , <-20° C, and ultra-low (<-60°C),
- Pre-validated against International Standards (ISTA 7D/ WHO)
- Easier to use and faster to assemble - as less as in 5 minutes
Esteemed Clients





























Collaborate With Us

We believe in creating impactful innovations through
collaboration and strategic partnerships.


