
Breaking the Ice
The use of ice as a food preservation technique dates back to 1000 BC dates and by 400 BC, Persian society had mastered the art of storing ice below ground level in desserts. The revolution really began in 1820, when an American businessman, Frederic Tudor – who was aka – Ice King of the World – developed a way to insulate ice from melting aboard the ships by using sawdust and put in place an effective national supply chain, distributing it as a commodity from New England to the hotter parts of the country and eventually to the world, rest is history!
Fast forward to 2024, in 224 years not only have we leap frogged in the supply chain efficiency of moving ice but we also have sophisticated ice-making machines, household refrigerators that can enable making ice at source. Hyper delivery ecommerce which delivers ice cubes in under 10 minutes avoids even the need for insulation to ensure you access to a bag of ice at a blink. That’s indeed a big leap forward!

Evolution of PCM
The evolution from Ice to PCM was led by the need for passive temperature control at various other temperatures other than °0 C. Different applications and products have varying requirements of temperature control. The three core attributes of Ice which may seem so natural yet exceptional are ;
- Ability to maintain a constant temperature,
- Ability to repeat the cycle of phase change at the same temperature point of 0°C
- Ability of retaining high thermal energy within a 6 times smaller volume in comparison to water.
Note that the thermal energy gained by water for every 0°C is a fraction of the energy gained the moment water reaches 0°C. To explain further, a bowl of water absorbs approximately 4KJ/ kg-K of thermal energy for every °C drop from say ambient temperature. The moment that the temperature crosses below +1°C transitioning into to 0 deg C the gain of thermal energy is 334 KJ/Kg-k and that is the very point phase starts to change from liquid to solid state. Subsequently, once the entire mass water is converted to ice, it further starts dropping in temperature with an energy gain at the rate of 2 Kj/kg-k for every °C drop in temperature. The graph below illustrates the energy gain in relation to temperature.
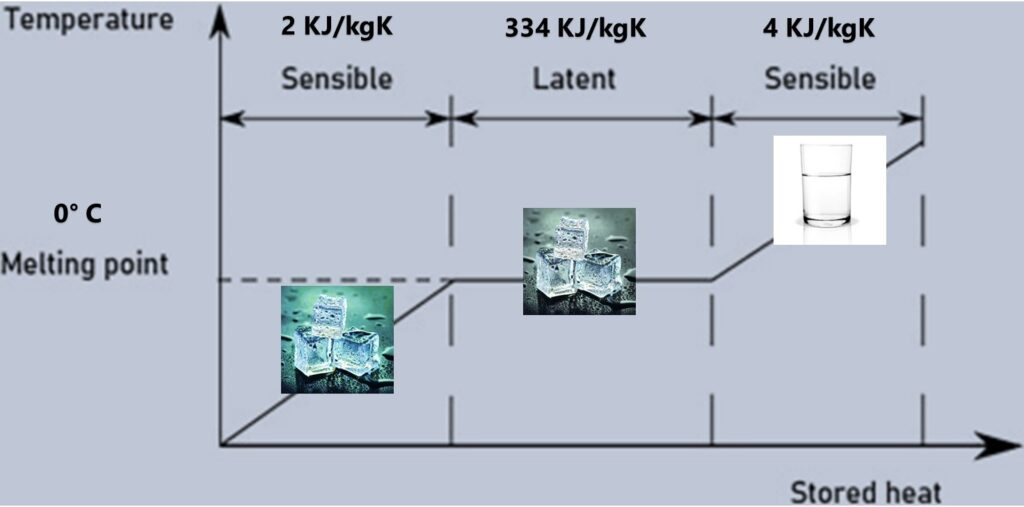
Phase Change Materials (PCM) or latent heat storage material is the most efficient ways to store thermal energy. The high energy storage per unit mass during phase change from solid to liquid and vice versa happens at a constant temperature. Essentially, PCMs emulate the property of water at different temperatures, In a layman’s term we can say PCMs are ice at different temperatures.
Versatility of PCM over ICE as a thermal energy storage medium
Like ICE, PCMs provide the ability to decouple the time of consumption of energy from the time of generation. For example, a stationary fridge which generates coolth actively to keep the content inside cold vs an insulated box which keeps the content inside cold with the help of ice cubes or packaged ice (commonly known as gel packs or Ice packs). Since, this approach does not require any external source of energy being needed at the time of use, this strategy is known as passive cooling.
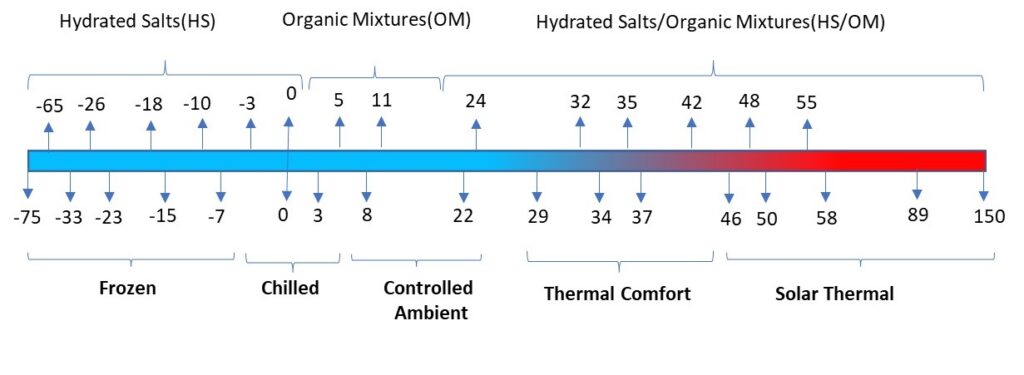
Each Phase Change Material (PCM) is tailored in its chemistry to ensure that it changes its phase at a designed temperature point, which in other words is the melting / freezing point. An ideal PCM can undergo several thousand cycles of melting and freezing demonstrating repeatability and stability of performance over yeas of use. The next important parameter is the measurement of its thermal energy storage capacity, which also varies for different PCMs. The figure below shows the range of PCMs available in the portfolio of offerings by Pluss Advanced Technologies.
What should be the key factors of selecting the right PCM?
Once the objective or the “why” of using PCM is understood the selection of the PCM is determined by following parameters listed in the order of priority;
- Temperature to be maintained – A ΔT of minimum 5°C to be maintained between the PCM & medium that needs to be heated or cooled.
- Temperature available for charging – A ΔT of minimum 5°C to be maintained between the PCM & medium used to heat or cool PCM.
- The surface of heat-exchange – Depending on demands of the application the compatibility of PCM with the surface that it comes in contact with should be checked.
- Weight & Volume – To determine whether the weight & volume added by the PCM would be feasible.
In summary PCM as an energy storage medium can be exploited in any field or application which requires heating or cooling. Storage of energy, in this case in the form of ‘thermal’ offers a plethora of applications which enable one to make things convenient for consumer products, to move away from dependency on fossil-fuel based generators, to bring in efficiencies of managing demand side response.
